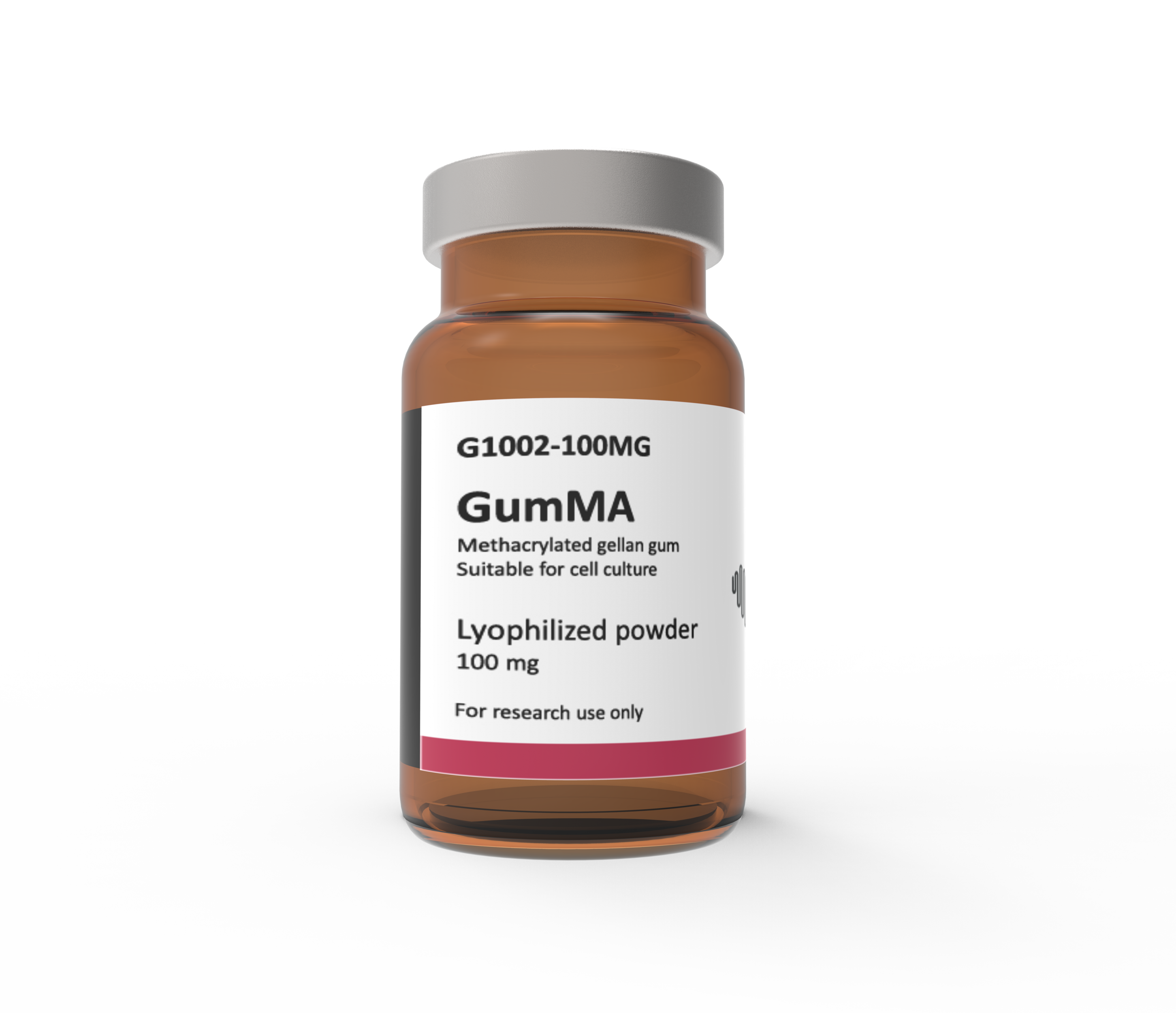
GumMA (Methacrylated Gellan Gum)
$120,00 – $590,00
Methacrylated Gellan Gum, which has FDA approval as a food additive, is an extracellular microbial anionic heteropolysaccharide consisting of glucose–glucuronic acid–glucose–rhamnose as a repeating unit and that forms a gel in the presence of metallic ions. Gellan Gum has a free carboxylic group per repeating unit, which can be used for functionalization and enhance its bio-stability. Methacrylated Gellan Gum is a new biomaterial for cartilage-tissue engineering fields due to its good mechanical properties and promising results as therapy.
Photo-polymerization is an alternative method for hydrogel formation with increased structural and mechanical integrity. In this technique, polymers are modified with methacrylates that undergo free radical polymerization in the presence of a photo-initiator and upon exposure to UV light. This polymerization reaction induces the formation of covalent crosslinks between functional methacrylate groups along the backbone of the polymer chains. Chemical modification of gellan gum (GG) with methacrylate is feasible and allows obtaining both ionic- and photo-crosslinkable methacrylated gellan gum hydrogels.
Studies have shown that gellan gum hydrogels are non-cytotoxic biocompatible materials.
Document



Reviews
There are no reviews yet.